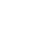Calcium oxide, commonly known as quicklime, undergoes a fascinating and significant reaction when it comes into contact with water. This reaction, known as slaking or hydration, is fundamental to numerous industrial processes, construction applications, and even environmental remediation. Understanding what happens when calcium oxide reacts with water is crucial for optimizing these processes and ensuring safety and efficiency. The resulting calcium hydroxide, or slaked lime, possesses entirely different properties than its parent compound, impacting its utility across diverse sectors.
Globally, the production and utilization of calcium oxide and its hydrated form represent a multi-billion dollar industry, underpinning critical infrastructure development, agricultural practices, and manufacturing processes. Fluctuations in calcium oxide supply chains can directly affect construction costs, wastewater treatment efficiency, and even the production of certain steel alloys. The ability to accurately predict and control what happens when calcium oxide reacts with water is, therefore, a key concern for industries worldwide.
The importance of comprehending this chemical process extends beyond purely economic considerations. It plays a vital role in carbon capture technologies, sustainable building materials, and the stabilization of contaminated soils. Furthermore, understanding the heat generated during the reaction is essential for safety protocols in industrial settings.
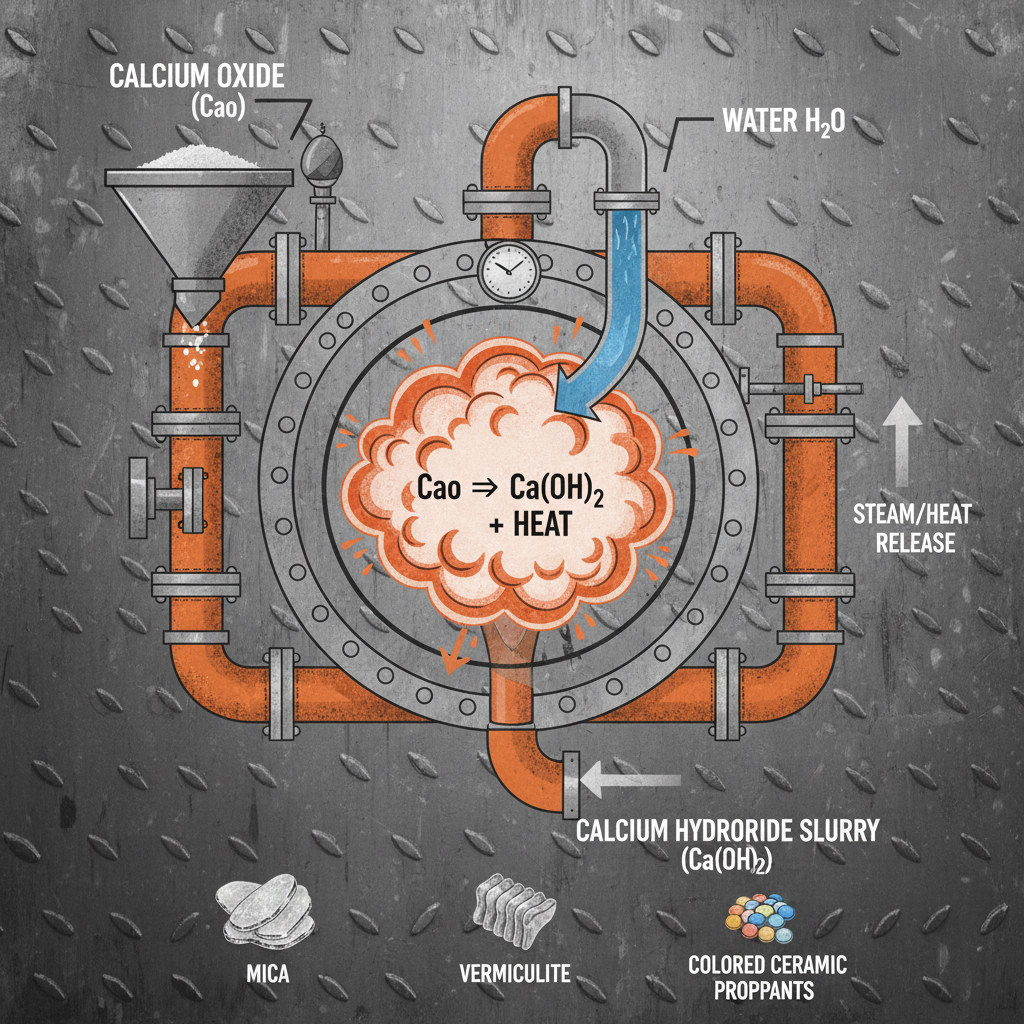
The reaction of calcium oxide with water, resulting in calcium hydroxide, isn't merely a chemical equation—it's a cornerstone of numerous industrial applications. Understanding this process is paramount because the properties of the resulting calcium hydroxide differ significantly from those of the original calcium oxide. This transformation dictates its use in everything from pH adjustment in wastewater treatment to the creation of durable building materials. What happens when calcium oxide reacts with water dictates the qualities and functionality of a wide range of products.
This reaction is highly exothermic, meaning it releases substantial heat. Managing this heat release is crucial for safe and efficient operations in industrial settings. The amount of heat generated depends on the purity of the calcium oxide and the rate of water addition, demanding careful process control.
What happens when calcium oxide reacts with water is a chemical reaction represented by the equation CaO (s) + H₂O (l) → Ca(OH)₂ (s) + Heat. Essentially, solid calcium oxide (quicklime) combines with liquid water to produce solid calcium hydroxide (slaked lime) and releases heat. This isn’t simply a dissolving process; it’s a chemical change where new bonds are formed. The quicklime's crystalline structure breaks down, and the calcium ions combine with hydroxide ions derived from the water.
The resulting calcium hydroxide is a white powder with alkaline properties. It's widely used in construction as a component of mortar and plaster, due to its binding properties and ability to set over time. Furthermore, it's a key ingredient in many industrial processes requiring pH control or neutralization. The efficiency of this process has direct humanitarian applications, particularly in water purification systems.
The speed and completeness of the reaction are influenced by several factors, including the surface area of the calcium oxide, the temperature of the water, and the rate of mixing. More finely ground calcium oxide reacts more quickly due to the increased surface area exposed to the water.
Several key factors directly impact the rate and efficiency of what happens when calcium oxide reacts with water. Firstly, particle size plays a crucial role: finer particles exhibit a larger surface area, leading to a faster reaction rate. Secondly, the temperature of the water influences the reaction speed – warmer water generally accelerates the process.
The purity of the calcium oxide is also critical. Impurities can inhibit the reaction, reducing the yield of calcium hydroxide. Furthermore, the mixing method significantly affects the reaction. Efficient mixing ensures uniform contact between the calcium oxide and water, promoting a complete and controlled reaction. Controlling the rate of water addition is crucial for managing the heat generated.
Finally, the presence of certain additives or catalysts can either accelerate or decelerate the reaction. Understanding these factors allows for optimized control of the hydration process, maximizing efficiency and ensuring product quality.
The impact of what happens when calcium oxide reacts with water resonates across numerous global industries. In construction, calcium hydroxide is an essential component of mortars and plasters, providing binding properties and contributing to the durability of structures. It is also extensively used in soil stabilization, improving the load-bearing capacity of foundations.
In the environmental sector, calcium hydroxide is vital for wastewater treatment, where it neutralizes acidic effluents and aids in the removal of heavy metals. It is also used in flue gas desulfurization, reducing sulfur dioxide emissions from power plants. Furthermore, in agriculture, it's employed to adjust soil pH and provide essential calcium nutrients for plant growth.
Utilizing calcium hydroxide derived from what happens when calcium oxide reacts with water offers significant advantages. Cost-effectively, calcium oxide is relatively abundant and inexpensive compared to many alternative materials. Sustainability benefits include its potential for carbon capture and utilization, reducing the environmental impact of industrial processes.
From a societal perspective, the use of calcium hydroxide in wastewater treatment contributes to public health and environmental protection. Its reliability as a stabilizing agent in construction enhances the safety and longevity of infrastructure. This holistic benefit profile makes it a compelling choice for various applications.
Ongoing research focuses on optimizing the hydration process for enhanced efficiency and sustainability. Nanotechnology is being explored to create calcium oxide nanoparticles with increased reactivity, leading to faster and more complete hydration. The integration of digital monitoring and control systems promises to automate and refine the process.
Furthermore, researchers are investigating the use of alternative water sources, such as recycled water, to minimize environmental impact. Innovations in carbon capture technologies leverage the exothermic nature of the reaction to sequester CO₂, creating a closed-loop system.
These advancements aim to improve the economic viability and environmental performance of calcium hydroxide production and utilization.
Despite its benefits, several challenges exist in effectively managing what happens when calcium oxide reacts with water. The significant heat release necessitates robust cooling systems and careful process control to prevent runaway reactions. Dust generation during handling poses health and safety concerns, requiring appropriate ventilation and personal protective equipment.
One solution is to implement advanced mixing technologies that promote more uniform contact between calcium oxide and water, reducing localized heat buildup. Improved dust control measures, such as enclosed handling systems and wet suppression techniques, can mitigate health risks.
Another challenge is ensuring consistent quality of the calcium oxide feedstock. Rigorous quality control procedures and sourcing from reputable suppliers are essential for maintaining optimal hydration performance.
| Particle Size (µm) | Water Temperature (°C) | Mixing Intensity (RPM) | Hydration Efficiency (%) |
|---|---|---|---|
| 10 | 25 | 500 | 95 |
| 50 | 25 | 500 | 80 |
| 10 | 60 | 500 | 98 |
| 10 | 25 | 200 | 70 |
| 30 | 40 | 300 | 65 |
| 20 | 35 | 400 | 78 |
Handling calcium oxide and water requires caution due to the exothermic reaction and the alkaline nature of calcium hydroxide. Always wear appropriate personal protective equipment (PPE), including safety glasses, gloves, and a dust mask. Ensure adequate ventilation to prevent inhalation of dust. Control the rate of water addition to manage the heat generated and avoid splashing. Promptly clean up any spills and avoid contact with skin and eyes.
The speed of the reaction can be controlled by adjusting several factors. Increasing the water temperature generally accelerates the reaction, while decreasing it slows it down. Controlling the particle size of the calcium oxide – finer particles react faster – and the rate of water addition are also crucial. Effective mixing ensures uniform contact and promotes a complete reaction at a controlled rate.
The theoretical stoichiometric ratio of calcium oxide to water for complete hydration is 1:1 (CaO + H₂O → Ca(OH)₂). However, in practice, a slight excess of water is often used to ensure complete reaction and to form a workable paste. The exact ratio may vary depending on the desired consistency and application. Careful monitoring is key to achieving optimal hydration.
Yes, recycled water can often be used for hydrating calcium oxide, provided it meets certain quality standards. It’s crucial to ensure the water is free from contaminants that could interfere with the reaction or compromise the quality of the resulting calcium hydroxide. Monitoring the pH and conductivity of the recycled water is recommended.
Calcium hydroxide produced from what happens when calcium oxide reacts with water offers several environmental benefits. It’s used in wastewater treatment to neutralize acidity and remove pollutants. It can also contribute to carbon capture and utilization. Its use in sustainable construction materials reduces the environmental footprint of buildings, and its ability to stabilize soils prevents erosion.
The substantial heat released during the hydration of calcium oxide can be both a benefit and a challenge. It requires effective heat management systems to prevent overheating and potential hazards. However, this heat can also be harnessed for other processes, such as preheating materials or generating steam, improving overall energy efficiency. Careful process control is essential to optimize heat utilization.
In conclusion, what happens when calcium oxide reacts with water is a foundational chemical process with far-reaching implications across numerous industries. Understanding the factors influencing the reaction, optimizing the hydration process, and leveraging the resulting calcium hydroxide are vital for achieving sustainable and efficient outcomes in construction, environmental remediation, and beyond. Its ongoing relevance is assured by continued innovations aimed at enhancing its performance and minimizing its environmental impact.
Looking ahead, further research into nanotechnology, process automation, and alternative water sources promises to unlock even greater potential for this versatile reaction. By embracing these advancements and prioritizing safety and sustainability, we can harness the power of calcium oxide hydration to address critical global challenges and build a more resilient future.


If you are interested in our products, you can choose to leave your information here, and we will be in touch with you shortly.