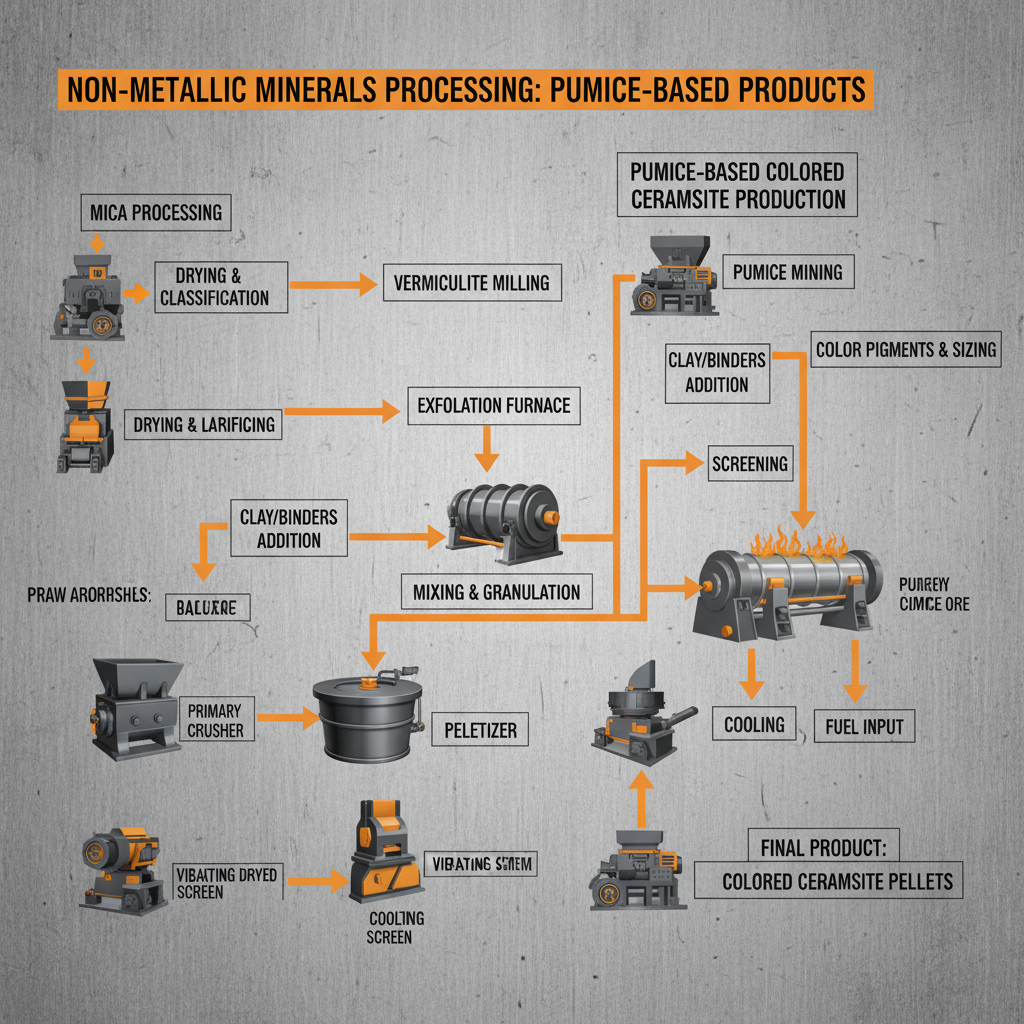
Understanding Calcium Oxide in Water: A Vital Component for Global Industry and Environment
Calcium oxide in water, often referred to as quicklime when dry, plays a surprisingly significant role in many industries and environmental applications worldwide. Its interaction with water forms calcium hydroxide, a compound central to water treatment, construction, and even humanitarian efforts. If you've never thought about the chemistry behind clean water or sustainable building materials, that’s okay — but frankly, having a handle on calcium oxide in aqueous conditions is an increasingly global concern.
Why does this matter? Globally, billions lack access to clean, safe water, and infrastructure demands are ever-growing. Understanding how calcium oxide reacts in water unlocks practical solutions that directly impact public health, industrial efficiency, and eco-friendly development. In this article, we explore the what, how, and why of calcium oxide in water, linking science to real-world benefits and innovations.
Global Context: The Widespread Relevance of Calcium Oxide in Water
As of recent UN water reports, nearly two billion people remain without safely managed drinking water services. Industrial and municipal water treatment plants worldwide rely heavily on chemical processes to remove contaminants. Calcium oxide in water creates calcium hydroxide, a base that helps neutralize acidity and precipitate heavy metals. Remarkably, the global production of quicklime exceeds 300 million tonnes annually (ISO, 2022), underscoring its ubiquity.
Yet, challenges persist: water sources contaminated with industrial runoff, acidic lakes, and inefficient sanitation systems need cost-effective and scalable solutions. The use of calcium oxide in water is often a front-line response.
Definition & Meaning: What Is Calcium Oxide in Water?
In simple terms, calcium oxide (CaO) is a white, caustic, alkaline crystal known as quicklime. When slaked with water, it forms calcium hydroxide (Ca(OH)₂), commonly called slaked lime. This reaction is exothermic, producing heat and a strong alkaline solution valuable for various industrial reactions.
Modern industries—from construction to agriculture and water treatment—depend on this chemistry. Humanitarian organizations also utilize calcium oxide in water for sanitation, neutralizing acidic soils, or safely treating drinking water in emergency contexts.
Key Factors Affecting Calcium Oxide’s Use in Water
1. Reactivity and Purity
The purity of calcium oxide dramatically influences its reaction with water—pure quicklime ensures predictable calcium hydroxide concentration and pH levels. Low-purity products may introduce impurities affecting water quality or downstream processes.
2. Scalability for Industrial Applications
Scalability is crucial: small-scale water treatment plants might add calcium oxide manually, while large municipal plants rely on automated dosing systems. The ability to customize quantities directly impacts cost-efficiency and treatment precision.
3. Cost Efficiency and Availability
Quicklime is relatively inexpensive compared to alternative chemicals, and its ready availability makes it a first choice in many regions. However, transport costs can escalate in remote areas, necessitating local processing facilities.
4. Environmental Impact and Sustainability
While calcium oxide treatment improves water safety, mining and producing quicklime require significant energy, often from fossil fuels. Innovations in sustainable sourcing and "green lime" production are emerging to address this concern.
5. Safety Considerations
Handling calcium oxide requires care — it's caustic and can cause skin or respiratory irritation upon contact. Proper training and personal protective equipment are essential, especially in large-scale applications.
Calcium Oxide in Water: Product Specification at a Glance
| Specification | Typical Value |
|---|---|
| Chemical Formula | CaO |
| Purity | ≥ 95% |
| Moisture Content | ≤ 1% |
| Particle Size | #80 mesh (average 180 μm) |
| pH of 1% Slurry | ~12.4 |
Global Applications & Use Cases of Calcium Oxide in Water
The utility of calcium oxide dissolved or reacted in water crosses many regions and industries. Here are some vivid examples:
- Water Treatment Plants: Across Asia and Africa, municipal systems depend on lime softening to remove hardness and precipitate heavy metals.
- Post-Disaster Relief: Emergency sanitation in places like Haiti or the Philippines often uses quicklime to treat water or latrines, neutralizing pathogens rapidly.
- Industrial Zones: Factories in remote locations, like mining operations in South America, use calcium oxide in water to stabilize acidic effluents before discharge.
- Construction & Soil Stabilization: In regions prone to flooding, adding calcium oxide to water for soil amendment strengthens foundations and prevents erosion.
These cases reflect why calcium oxide is sometimes called the "backbone" for sustainable water and soil management. If you’d like to dig deeper, check out calcium oxide in water applications globally.
Advantages & Long-Term Value of Calcium Oxide in Water Applications
The appeal of calcium oxide in water isn’t just technical — it’s also physical and emotional. Here’s why:
- Cost-Effectiveness: Affordable raw material with well-understood chemistry makes it reliable for even the most budget-conscious projects.
- Environmental Impact: When used carefully, it reduces water toxicity and supports ecosystem balance.
- Social Impact: Enhancing access to safe water fosters dignity, health, and improved quality of life.
- Durability & Reliability: Calcium hydroxide formed helps maintain stable water conditions for long durations.
- Innovation Driver: Emerging "green lime" tech is turning calcium oxide applications into cleaner, carbon-neutral solutions.
Honestly, it feels like calcium oxide in water is one of those unsung heroes — quietly making a measurable difference to millions.
Future Trends & Innovations
The landscape is shifting quite a bit. Green manufacturing techniques aim to cut CO₂ emissions associated with lime production—think renewable power and carbon capture. Digital sensors now enable precise dosing of calcium oxide slurries, optimizing efficiency and reducing waste. In addition, some companies are exploring combining calcium oxide with bio-based additives to enhance water purification while minimizing ecological footprints.
Challenges & Solutions in Using Calcium Oxide in Water
Of course, no technology is perfect. One big hurdle is the handling and storage of quicklime — it's caustic and exothermic, requiring rigorous safety protocols. Transport logistics, especially in remote areas, can hike costs.
Innovative approaches involve on-site slaking facilities to generate calcium hydroxide locally, reducing risks and transfer inefficiencies. Training programs and standardized protocols also help mitigate user errors and ensure safe handling.
FAQ: Frequently Asked Questions About Calcium Oxide in Water
- How does calcium oxide help in water purification?
- When calcium oxide dissolves in water, it forms calcium hydroxide, which neutralizes acidic water and precipitates impurities such as heavy metals, making water safer for consumption or industrial use.
- Is calcium oxide safe to handle?
- Calcium oxide is caustic and can cause skin or eye irritation. Proper protective equipment and training are essential during handling. Once slaked into calcium hydroxide and diluted, it is much safer.
- Can calcium oxide be used in emergency water treatment?
- Yes, humanitarian groups often use quicklime in disaster zones to treat water or improve sanitation due to its effectiveness against pathogens and affordability.
- What industries benefit most from calcium oxide in water?
- Water treatment, construction, mining, and agriculture are key industries that leverage calcium oxide’s water reactivity for purification, soil stabilization, or pH control.
- How can I ensure the quality of calcium oxide for water applications?
- Look for products with high purity (≥ 95%), low moisture, and appropriate particle size. Certifications from reputable suppliers and ISO standards help guarantee quality.
Vendor Comparison: Leading Calcium Oxide Suppliers
| Vendor | Purity | Price (USD/ton) | Availability | Certifications |
|---|---|---|---|---|
| Baifeng Mining | ≥ 98% | $120 | Global | ISO 9001, ISO 14001 |
| LimePro Inc. | ≥ 95% | $110 | North America | ISO 9001 |
| EcoLime Global | ≥ 96% | $130 | Europe, Asia | ISO 14001, GreenCert |
Conclusion: The Lasting Impact of Calcium Oxide in Water
Calcium oxide in water is more than just a chemical reaction—it’s a cornerstone of sustainable water treatment and environmental protection. Its affordability, versatility, and global applicability are unmatched for many developing regions and industrial sectors alike. By investing in high-quality calcium oxide and advancing greener production methods, we pave the way for cleaner water, healthier communities, and a more resilient planet.
Curious to learn more or source quality calcium oxide? Visit our website: https://www.baifengmining.com for cutting-edge solutions.
Mini takeaway: Calcium oxide’s interplay with water quietly supports global clean water initiatives and industrial sustainability – worth a closer look!
References
Related News