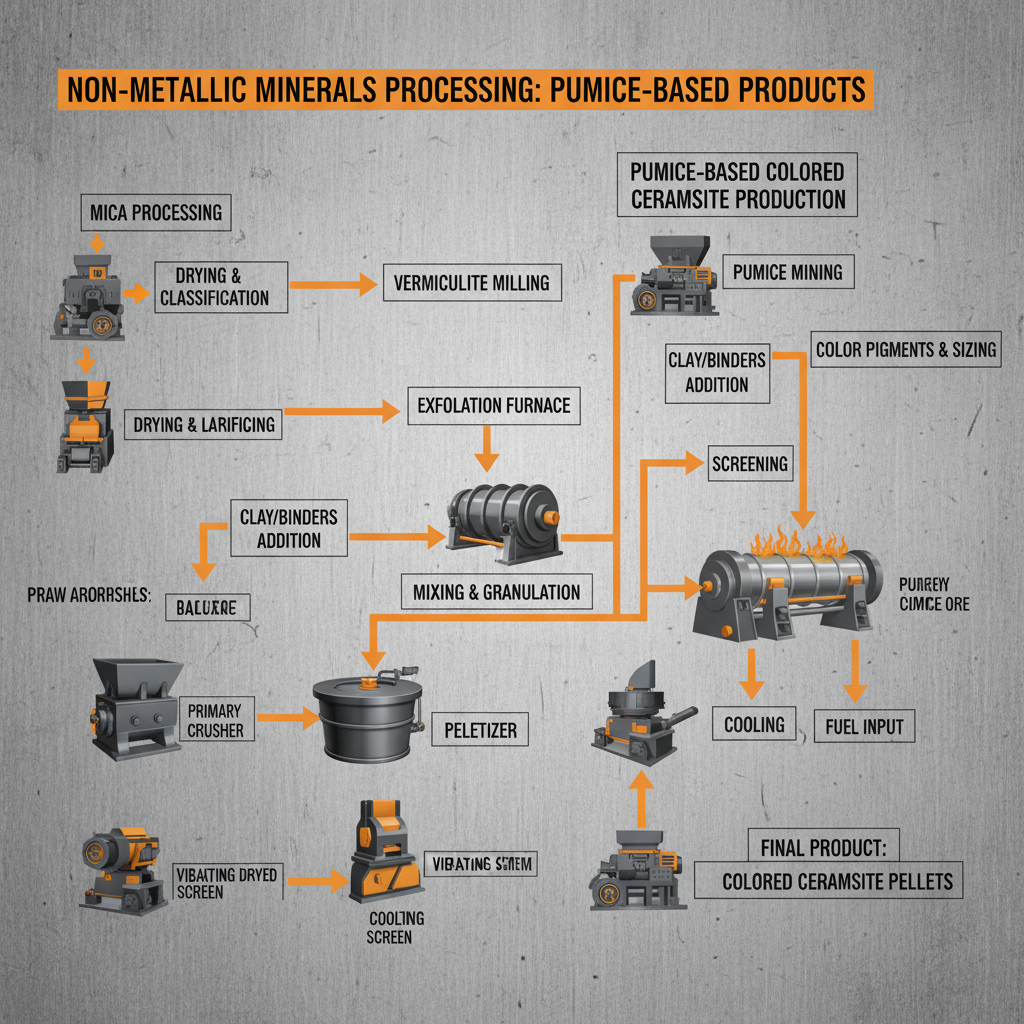
The Essential Role of Ca(OH)₂ and H₂SO₄ in Global Industry and Environment
Understanding Ca(OH)2 and H2SO4: Why it Matters Globally
If you’ve ever dabbled in chemistry or industrial processes, the compounds calcium hydroxide (Ca(OH)2) and sulfuric acid (H2SO4) likely ring a bell. But beyond their simple chemical formulas, these two substances are cornerstones in industries shaping the world’s infrastructure, environmental safety, and even humanitarian efforts. This article dives into the global relevance of ca oh 2 h2so4, their connection, and why understanding their interaction can ripple across sectors from mining to water treatment, with benefits that aren’t immediately obvious to the casual observer. ---The Global Stage: Why Ca(OH)2 and H2SO4 Matter
Ca(OH)2, commonly known as slaked lime, and sulfuric acid, a strong mineral acid, are foundational in many large-scale industrial applications worldwide. According to the International Energy Agency and United Nations reports, sulfuric acid production alone surpasses 260 million tonnes annually, mostly used in fertilizer manufacturing—critical for food security globally. Meanwhile, calcium hydroxide plays a pivotal role in neutralizing acidic ores and purifying water—a task especially pressing in regions afflicted with acid mine drainage or water pollution. But here’s the kicker: the interaction between ca oh 2 and h2so4 doesn’t just produce a simple reaction — it’s a linchpin in environmental remediation and resource efficiency. Consider that as industry expands, managing acidic byproducts efficiently is more than a technical challenge; it’s an environmental necessity. ---Ca(OH)2 and H2SO4 Defined in Plain Terms
Simply put: calcium hydroxide is an alkaline substance, while sulfuric acid is strongly acidic—their union results in calcium sulfate (aka gypsum) and water. This reaction (Ca(OH)2 + H2SO4 → CaSO4 + 2H2O) is not just a textbook chemical equation but a fundamental process in industries across the board. This isn’t just about chemistry homework. When industrial waste containing sulfuric acid hits the environment, calcium hydroxide is often the material used to neutralize the acidic content, preventing soil degradation and water toxicity. This makes the pair critical in sustainable industrial practices and in meeting ISO environmental standards. ---Core Factors Behind Effective Ca(OH)2-H2SO4 Use
1. Purity of Calcium Hydroxide
Impurities in Ca(OH)2 can hamper the neutralization efficiency, leading to incomplete reactions and residual acidity in waste streams.2. Concentration of Sulfuric Acid
The amount and strength of H2SO4 guides dosing—the acid’s molarity impacts how much Ca(OH)2 is needed, which affects cost and reaction time.3. Reaction Conditions (Temperature & Mixing)
Faster and more complete reactions happen at optimal temperatures; insufficient mixing can cause uneven neutralization, presenting operational hazards.4. By-product Management
The gypsum formed can be a waste product or a resource—industries increasingly explore reusing calcium sulfate for construction materials, circular economy style.5. Cost Efficiency
Balancing raw material costs, logistics, and environmental compliance shapes the attractiveness of Ca(OH)2 and H2SO4 processes in industry.Mini takeaway:
It often feels like the success of this chemistry hinges on neat details — purity, dosage, and process control. When those align, you get a reliable, sustainable solution. ---Real-World Applications: Where Ca OH2 Meets H2SO4
Industrially, these substances touch many sectors: - Mining: Acid mine drainage is an environmental nightmare. Ca(OH)2 neutralizes acidic effluents containing sulfuric acid, protecting freshwaters and ecosystems. - Water Treatment Plants: Hard water treatment uses these compounds for adjusting pH; safer drinking water follows. - Fertilizer Manufacturing: The sulfur content in fertilizers often traces back to sulfuric acid processing. - Construction: Gypsum by-products become drywall and cement additives—closing the waste loop. - Disaster Relief Logistics: In areas where water acidity spikes due to contamination, rapid deployment of neutralizing agents such as Ca(OH)2 can safeguard communities. In particular, countries rich in mining activities—think Chile, Australia, South Africa—lean heavily on this chemistry combo. NGOs operating in post-industrial pollution zones also value Ca(OH)2 treatment protocols. ---Advantages & Long-Term Value of Ca(OH)2 and H2SO4 Technologies
When properly managed, the reaction between calcium hydroxide and sulfuric acid provides several major benefits: - Cost efficiency: Both chemicals are relatively inexpensive and effective on a large scale. - Sustainability: Treating waste acid streams reduces soil and water pollution sustainably. - Social Impact: Cleaner environments promote healthier communities downstream of industrial zones. - Reliability: The reaction is predictable, helping industries meet environmental standards. - Innovation: New approaches in by-product re-use are turning waste gypsum into valuable materials. There’s something reassuring about knowing that a simple chemical neutralization process contributes to global efforts for sustainability and safety — in a world increasingly conscious of ecological footprints. ---Future Trends & Innovations in Ca(OH)2-H2SO4 Handling
Looking ahead, a few exciting roads appear: - Green Chemistry: Researchers are developing bio-based alternatives that could supplement or replace synthetic Ca(OH)2 to lessen mining demands. - Automation & Sensor Integration: Automated dosing systems with real-time pH sensors optimize neutralization, saving resources. - Recycling By-products: Turning gypsum waste into building materials or as a filler in industrial products gains momentum. - Energy Efficiency: Improved reaction vessel designs cut energy overhead in large-scale neutralization plants. - Policy & Compliance: Tighter soil and water quality policies push industries to adopt better Ca(OH)2 dosing and treatment protocols. Frankly, the future looks greener and smarter, riding on digital transformation and circular economy principles. ---Challenges & Solutions in Current Practice
No process is perfect. Some persistent challenges include: - Inconsistent quality of raw materials causing unpredictable results. - Handling large volumes of sludge or gypsum waste. - Managing reaction heat in large batch processes. - Ensuring compliance with ever-stricter environmental laws. Solutions flow from better quality control, modular treatment units to handle volume variability, and stronger collaboration between chemical suppliers and end-users. Some companies are experimenting with real-time analytics and AI to anticipate process hiccups before they become headaches. ---FAQ: Frequently Asked Questions About Ca OH2 H2SO4
Q1: How does calcium hydroxide neutralize sulfuric acid in industrial effluents?A1: Calcium hydroxide reacts with sulfuric acid to form calcium sulfate and water, effectively neutralizing acidity. This process helps prevent environmental harm from acidic waste, especially in mining runoff or wastewater treatment.
Q2: Can the gypsum produced from this reaction be reused?
A2: Yes, the by-product gypsum can be repurposed in construction materials such as drywall, cement additives, or soil conditioners. This recycling reduces waste disposal costs and supports circular economy goals.
Q3: What are common challenges in dosing Ca(OH)2 for acid neutralization?
A3: Challenges include ensuring proper mixing, maintaining purity of calcium hydroxide, and accurately measuring acid concentrations to avoid under- or overdosing that reduces efficiency or creates hazardous conditions.
Q4: Is this chemical neutralization method environmentally safe?
A4: When managed correctly, it’s considered environmentally sound since it neutralizes harmful acidic compounds. However, by-product disposal and raw material sourcing should be handled sustainably.
Q5: How do industries source and transport Ca(OH)2 and H2SO4 safely?
A5: Safety protocols involve using sealed containers, temperature controls, and trained handling teams, as sulfuric acid is highly corrosive, and calcium hydroxide is caustic. Compliance with UN transport regulations ensures risk mitigation. ---
Product Specification Table for Ca(OH)2 and H2SO4
| Specification | Calcium Hydroxide | Sulfuric Acid |
|---|---|---|
| Chemical Formula | Ca(OH)2 | H2SO4 |
| Appearance | White powder/paste | Colorless to slightly yellow liquid |
| Purity | >95% | >98% |
| Density | ~2.2 g/cm³ (solid) | 1.84 g/cm³ (liquid at 20 °C) |
| pH (1% solution) | ~12 (alkaline) | ~1 (acidic) |
| Common Uses | Neutralization, water treatment, soil stabilization | Fertilizer production, mineral processing, pH adjustment |
Vendor Comparison Table: Ca(OH)2 and H2SO4 Suppliers
| Vendor | Purity (%) | Capacity (tons/month) | Price Range (USD/ton) | Global Shipping | Sustainability Initiatives |
|---|---|---|---|---|---|
| Alpha Chemicals Ltd. | >98 (H2SO4) | 25,000 | $150 - $200 | Yes | Improved waste recycling |
| Beta Minerals Inc. | >95 (Ca(OH)2) | 40,000 | $80 - $120 | Limited | Energy-efficient production |
| Gamma Industrial Supplies | >97 Both chemicals | 15,000 | $130 - $180 | Yes | Sourcing from sustainable mines |
In Conclusion: Why Ca(OH)2 and H2SO4 Remain Industry Pillars
From mitigating environmental damage to creating essential industrial outputs, the duo of calcium hydroxide and sulfuric acid defines a fascinating niche of chemistry with outsized impact. Understanding ca oh 2 h2so4 processes unlocks insights into resource optimization, sustainability, and technological progress. If your field touches mining, water treatment, or environmental management, diving deeper into these substances is no longer optional — it’s crucial. Curious for more details or looking for trusted suppliers? Visit our website: https://www.baifengmining.com and explore how this chemistry can power your projects sustainably. ---References
Related News


Copyright © 2026 Shijiazhuang Baifeng Mining Co.,Ltd All Rights Reserved. Sitemap | Privacy Policy | Top Blog | Global Service